Weeks of CMC drafting, done in minutes.
CMC Author transforms your technical source documents into structured, submission-ready regulatory content, with full traceability and zero hallucination.
Previously at
Regulatory submissions shouldn't take this long
Preparing regulatory submission content is one of the biggest bottlenecks in getting a drug to market.
Weeks of manual drafting
Regulatory specialists spend weeks translating batch records, analytical reports, and stability data into the structured prose and tables that agencies require.
Inconsistencies across sections
When the same data point appears in multiple places, manual entry leads to mismatches that aren't caught until review — then requires painful rework.
No traceability
Auditors need to know where every piece of content originated. In most workflows, that trail lives in someone's memory or scattered across emails.
Meet CMC Author
A purpose-built authoring tool for regulatory submission content. Upload your source documents and get structured, traceable, submission-ready drafts — organized around the CTD format. If the source document doesn't contain the information, the field stays blank. Nothing is fabricated.
Upload your source documents. Get a regulatory draft.
Drop in your technical source documents — batch records, analytical reports, stability data — and CMC Author generates structured content for the relevant CTD sections. Introduction text, process flow diagrams, narrative paragraphs, and supporting tables. Powered by structured extraction built on reliable NLP, not generative guesswork.
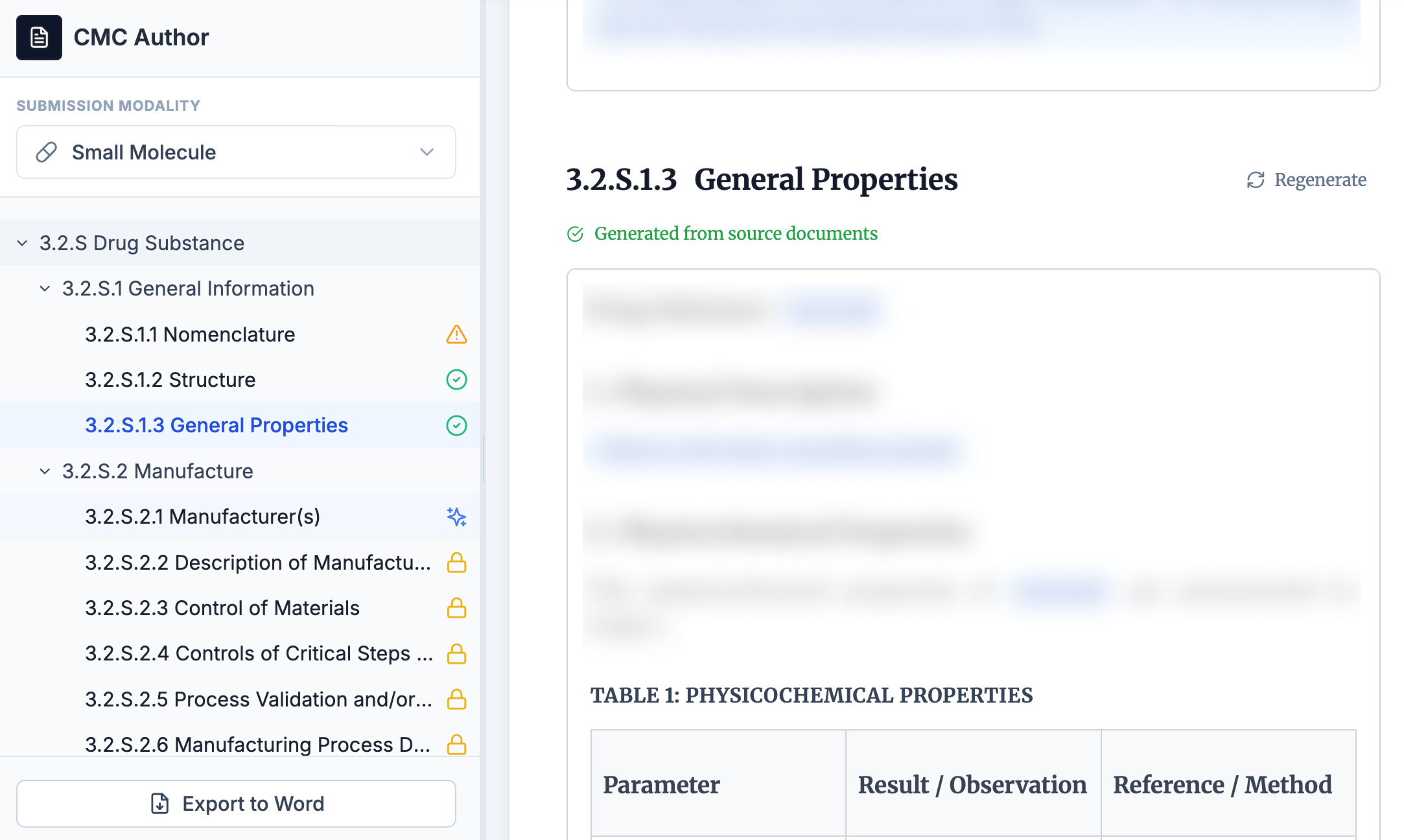
A structured workspace for every CTD section
A navigation panel tracks every section's completion status. A guidance panel delivers real-time regulatory context. Start any section from a template or from uploaded source documents. A built-in copilot suggests what to work on next.
The active ingredient is mixed with excipients. The mixture undergoes dry granulation wet granulation to form the final blend for compression.
Change it once. It updates everywhere.
Data entered in one place propagates to every location it appears — including flow diagrams. Change a process step in the narrative and the flow chart updates instantly. The system flags inconsistencies before they reach a reviewer.
Every sentence traced to its source
Traceability mode shows the origin of every piece of content — extracted, manually entered, or edited — along with the author and timestamp. Quality teams get the audit trail they need without adding overhead to the author's workflow.
How it works
Upload source documents
Batch records, analytical reports, stability data — drop them in and CMC Author handles the rest.
Review the generated draft
Structured content appears in CTD format, section by section, with source traceability on every element.
Fill gaps and refine
The system shows what's missing. Use smart autofill, manual entry, or the copilot to complete each section.
Export and submit
Export to Word in the format your agency requires — one continuous document for IMPD or separate files for FDA.
Built for regulated environments
Trust is earned, not claimed. Here's how we approach it.
No fabricated content
CMC Author uses structured extraction, not open-ended generation. If the information isn't in your source document, the field stays blank. You'll never find invented data in your submission.
Audit-ready by default
Every piece of content logs its origin — extracted, manually entered, or edited — along with the author and timestamp. Traceability is built into the workflow, not bolted on.
Enterprise-grade data handling
Your documents contain proprietary science and regulated data. We take that seriously. Contact us to learn about our security architecture and data handling practices.
Ready to accelerate your next submission?
Tell us about your team and we'll schedule a personalized walkthrough.